Sleep Medication Reviews for People With Bi Polar
Introduction
Bipolar disorder is a major health issue and cause of global disability (i). It is characterized by episodes of mania or hypomania and recurring depressive episodes that account for a considerable morbidity and mortality (2–4).
Depressive episodes, either in its major, dysthymic or mixed forms, are responsible for most of the morbidity in type I and type Ii bipolar patients at any stage of the disease progression and under whatever treatment (2, four). Current treatments are more effective managing manic episodes than in depressive ones. The utilise of antidepressants reduces symptoms of acute bipolar depression, notwithstanding, they do not increment clinical response or remission rates and have been associated with a long-term increased hazard of treatment-emergent mania or hypomania (5). Cognitive impairment in bipolar depression is associated with dysfunction and disability and a higher rate of premature decease when compared with the general population (6). This is caused by higher take chances of suicide, accidents (6) and multiple medical causes (7). This highlights the demand to explore effective treatment alternatives for acute bipolar depression.
Sleep deprivation (SD) is a treatment technique used for depression adult in the by fifty years (8, nine). Clinical studies have shown rapid antidepressive effects within 24–48 h later on SD and even during the same night or following morning (10, xi). Notwithstanding, interest in their clinical use has diluted because it soon became axiomatic that slumber recovery tended to contrary the clinical effect (12, thirteen). The best documented SD modality used with antidepressant purposes is Full sleep impecuniousness (TSD), though other modalities have been tested as shown in Table 1 (14, 15).

Table 1 Sleep deprivation modalities.
Historically, SD has shown response rates between fifty and eighty% (12) in a wide spectrum of depressive disorders, with no differences by gender or age (xiii, sixteen). However, these rates are twice as high in so called endogenous depressions than in neurotic depressions or secondary ones (xiii, 16). Bipolar patients take even ameliorate outcomes when compared to endogenous recurrent unipolar ones (17). Other variables associated with SD response are: diurnal variation of mood (13, fourteen, 18), high level of thyroid function, low peripheral sympathetic activity, an altered Dexamethasone suppression test (11, 12), among others (19–23). Nonetheless, despite the mentioned and well established SD antidepressant outcome, the clinical response to SD decreases apace (14). Relapse rates to SD are up to 83% after 1 night of sleep recovery (23); this has limited their clinical use. In club to avoid relapse, different SD schemes accept been created, and the use of medication (serotonergic drugs, lithium salts) or other chronotherapeutic therapies has been added (12). Studies demonstrate that a combined treatment tin can lead to sustained euthymia later on several months (12). However, the impact of the acute effect of SD combined with mood medications, besides as the long-term efficacy of TSD have simply been evaluated in clinical studies with a limited number of patients. This makes it difficult to obtain conclusive results. In our search, simply two previous meta-analysis about SD were institute (18, 24), and none of them directly addressed the long-term consequence of drug administration in combination with SD.
Therefore, we aimed to address a twofold research question: What are the acute effects of adding TSD to pharmacological handling? and, What are the astute and long-term effects of adding medications to TSD?
Methods
Search Strategy and Selection Criteria
In this systematic review and meta-analysis, nosotros searched MEDLINE, Embase, and the Cochrane Central Annals of Controlled Trials databases from their inception until October 28, 2019, using the search term "(sleep AND impecuniousness AND bipolar AND low)" with results filters based on report types (clinical trials and randomized controlled trials). We also searched ClinicalTrials.gov using the search term "(sleep AND impecuniousness AND bipolar AND depression)". We reviewed references and citations of articles retained in this study for additional unidentified studies.
Inclusion criteria were: clinical trials recruiting patients aged 18–75 years undergoing a depressive episode with a chief diagnosis of bipolar I or bipolar II disorder according to the electric current versions of the DSM or ICD at the time of the respective trial. Patients must have received combined medication treatment and sleep deprivation therapy (with or without additional chronotherapy, run into Tables 2 and 3). The existence of a control group was required.

Table 2 Characteristics of included trials in the starting time meta-assay.

Table 3 Characteristics of included trials in the second and tertiary analysis.
Exclusion criteria were: trials including inadequate samples (i.e. not exclusively bipolar patients or not control grouping) or interventions different from chronotherapeutic plus medication strategies. Studies with nonclinical outcomes (i.e. pharmacokinetics, serum concentrations, neuroimaging, etc.), or focused on clinical outcomes different from improvement, such as rubber/tolerability, were also excluded. Studies where information could not exist extractible or are non reported in raw class were excluded.
Information Analysis
For each published study, summary estimates were extracted independently by JPRM and ERS, with subsequent verification (by PAV), review, and consensus. The following variables were extracted in a structured manner: patient characteristics (mean age, sex, and primary diagnosis), medication used (dose and titration schedule), chronotherapy technique used, and placebo condition, treatment-emergent hypomania or mania episodes, and dropouts.
The master consequence was changed in clinician-rated depressive symptom score (Hamilton depression rating scale [HDRS] (31) or Montgomery–Åsberg Depression Rating Calibration [MADRS] (32), calculated as standardized mean differences (SMDs) (33). The secondary result was clinical response at approximately three months, episodes of treatment-emergent mania or hypomania, and tolerability. We used odds ratios (ORs, with 95% CIs). Clinical remission was defined past the written report's authors as HDRS-21 score of 7 or less or MADRS score of five or less. Nosotros used dropout rates as a proxy for tolerability, although they might too reflect the absenteeism of efficacy.
Since treatment effects are probable to vary between studies due to methodological differences—such every bit patient selection, primary diagnosis, medication used, and trial duration—nosotros used pooled random-furnishings models (34). Merely intention-to-care for data was analyzed (35). Meta-regression procedures were developed to examination the effect of time of handling on SMDs.
In lodge to assess heterogeneity betwixt trials (i.e. clinical and methodological multifariousness), Q statistics and I² were practical, with the threshold for heterogeneity divers as p value for the Q statistic of less than 0,one or I² greater than 35% (36). Publication bias was evaluated by inspection of funnel plots and Egger'due south exam. Quality cess was conducted with Cochrane Risk of Bias Tool for Randomized Controlled Trials (37). All analyses were performed using Stata 16.0 (StataCorp LP, College Station, TX, USA).
Results
Nosotros initially identified ninety potentially eligible studies (Figure 1). Removal of duplicates and screening of titles and abstract left ten records, of which iii were excluded afterwards full-text review considering of inadequate mixed samples (38–40). I included study did not report clinical improvement on previously defined rating scales, nevertheless it reported dropout rates and episodes of treatment-emergent mania or hypomania (30).

Figure one Study option. CENTRAL, Cochrane Fundamental Register of Controlled Trials.
Our meta-analysis included 311 bipolar patients with a depressive episode (fourscore with bipolar I disorder (DSM IV), 221 for whom the exact diagnosis was unspecified (DSM IV) and 10 with DSM-III-R bipolar disorder, Tables 2 and 3). Two trials included only type I bipolar patients. The mean age was 45.7 years (SD 4.2), and the overall sample consisted of 104 men and 170 women (the gender of 37 patients was not specified).
The 7 selected trials (11, 25–30) included patients that used TSD as a chronotherapy. Of these vii, iv used 3 cycles of TSD, one used three cycles of TSD plus light exposure (intensity of 150 or ii,500 lx), one used one bicycle plus vivid light and SPA, and i used three cycles of TSD plus three cycles of SPA (Tables 2 and three). All seven trials used mood medications. In 2 studies (xi, 25) both intervention and control groups received mood medication. There is no take chances for a sleep deprivation placebo condition (Table two). In the remaining five studies (26–thirty) both groups underwent TSD, only the intervention group received medication; of these v only two studies administered placebo to the control group (Table 3). The take chances of bias assessment was conducted in all included studies, see supplementary material (Figure S1 and Table S1).
Clinical Effectiveness of TSD Add-on to Mood Medication
Depressive symptom scores were available for 5 trials, two of them comparing the addition of TSD to mood medication (11, 25). Symptoms were measured with HDRS-17 and HDRS-nineteen weekly. Both groups were treated with medications (fluoxetine and antidepressant/mood stabilizer, respectively), and one of the groups received additional 3 cycles of TSD and one bicycle of TSD, brilliant light and SPA, respectively (Table 2). Compared with medication solitary, adding TSD was associated with a significant improvement in clinician-rated depressive symptoms afterwards 1 week (SMD −0.584 [95% CI −1.126 to −0.042], p = 0.03; Figure 2A). Between-sample heterogeneity was not significant, with a Q statistic of 0.04 (degree of liberty (df) = 1; p = 0.83) and a I2 of 0. Visual inspection of funnel plot showed asymmetry (Figure 2B).
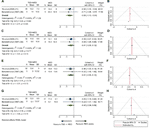
Effigy 2 Clinician-rated depressive symptoms adding TSD to mood medication and the funnel plot of included studies at the 1st calendar week (A, B), iind calendar week (C, D), 3rd calendar week (E, F) and 4th week (G, H). TSD, Total slumber deprivation; MED, Medication; Northward; Sample size; CI, Confidence interval; SD, Standard deviation. Tests of heterogeneity, I2 and Q statistic were included. REML, Restricted maximum likelihood.
The following weeks were associated with a nonsignificant tendency towards comeback in clinician-rated depressive symptoms: 2nd calendar week (SMD −0.413 [95% CI −0.949 to 0.124], p = 0.thirteen; Effigy 2C, for funnel plot encounter Figure second), threerd week (SMD −0.412 [95% CI −0.949 to 0.124], p = 0.13; Effigy 2E, for funnel plot see Figure 2F), and ivth calendar week (SMD −0.45 [95% CI −0.988 to 0.088], p = 0.1; Figure 2G, for funnel plot run into Figure 2H). To test the effect of fourth dimension equally a factor, we performed a meta-regression, that was nonsignificant (Coefficients 0.037 [95% CI −0.204 to 0.278], p = 0.765).
Clinical Effectiveness of Medication Added to TSD at Twenty-four hours 10
Four trials compared the addition of mood medication to TSD (26–29). Depressive symptoms were measured with HDRS-21 and MADRS at the 10th day. In three studies, patients were treated with three cycles of TSD, and in ane of them patients were treated with iii cycles of TSD and SPA. Mood medication (lithium, amineptine, and pindolol) was added to one group (Tabular array 3). Compared with TSD alone, adding mood medication was associated with a pregnant improvement in clinician-rated depressive symptoms after ten days of treatment (SMD −0.894 [95% CI −1.388 to −0.399], p < 0.001; Effigy 3A). There was evidence of heterogeneity betwixt the studies (I2 = 47.94, Q = v.76, df = 3, p = 0.12). Visual inspection of funnel plot showed asymmetry (Figure 3B), only Egger exam did non propose small-written report effects (β 1 = 2.59, SE of β 1 = 12.viii, z = 0.two, p = 0.84).
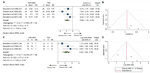
Figure 3 Clinician-rated depressive symptoms at the 10thursday twenty-four hours calculation medication to TSD (A) and the funnel plot of included studies (B). Clinician remission at the 3rd month (C) and the funnel plot of included studies (D). TSD, Total sleep impecuniousness; MED, Medication; CI, Confidence interval; SD, Standard departure; Due north, Sample size; R, Remission; NR, No remission; Tests of heterogeneity, Iii and Q statistic were included. REML, Restricted maximum likelihood.
Clinical Remission of Medication Added to TSD at 3 Months
Data for response rates were available for three trials that compared the addition of mood medication to TSD (26–28) (Tabular array 3). Clinical remission was divers by the report authors every bit HDRS-21 score < viii. Pooled log OR was 2.365 (95% CI 0.95 to three.779, p < 0.001; Figure 3C), compared with TSD, adding mood medication was associated with a meaning clinical remission subsequently 3 months. There was evidence of heterogeneity between the studies (Iii = 46.35, Q = 3.71, df = 2, p = 0.16). Visual inspection of funnel plot showed slight asymmetry (Figure 3D), however Egger test did non suggest modest-written report effects (β 1 = 6.71, SE of β 1 = 3.9, z = ane.72, p = 0.0854).
Tolerability and Treatment-Emergent Mania or Hypomania
The rates of treatment-emergent mania or hypomania were available for 4 studies (11, 27, 29, 30). At the end of the treatment, 5 (four.35%) of 115 patients given medication combined with TSD, four (4.17%) of 96 patients given TSD and cipher (0%) of 17 patients given mood medication had episodes of treatment-emergent mania or hypomania. Due to different control groups in the studies that reported affective switch (11, xxx), it was not possible to pool the data in a meta-analysis (Tables two and 3).
The rates of dropout were bachelor for all studies (11, 26–30). Overall, nine (5.8%) of 155 patients given mood medication combined with TSD, four (2.92%) of 137 given TSD and zero (0%) of 17 given mood medication were lost to follow-up (Tables ii and 3). Pooled log OR was 0.234 (95% CI −1.164 to ane.632, p = 0.74; Figure 4A) compared with TSD; adding mood medication was not associated with differences tolerability (dropout rate). Between-sample heterogeneity was not significant (Iii = 0, Q = 0.44, df = 1, p = 0.5). Visual inspection of funnel plot showed slight disproportion (Figure 4B).

Figure 4 Tolerability at the stop of the trial adding medication to TSD (A) and the funnel plot of included studies (B). TSD, Total sleep deprivation; MED, Medication; CI, Conviction interval; SD, Standard deviation; N, Sample size; D, Dropout; NR, No dropout; Tests of heterogeneity, Itwo and Q statistic were included; REML, Restricted maximum likelihood.
Discussion
This meta-assay provides three main results regarding the efficacy of Slumber deprivation (SD) in bipolar low. Our first result suggests that adding TSD to medication acutely increases the antidepressant effectiveness relative to mood medications alone at the commencement week, although this result was non maintained in the post-obit weeks. The second result shows that the addition of medication to TSD increased antidepressant effectiveness at day 10 of treatment relative to TSD lone. The third result yields the conclusion that the use of medication plus TSD increased clinical remission at a 3-month follow-upwardly. Finally, regarding tolerability and handling-emergent mania or hypomania, our results show no differences between treatments.
We initially proposed a twofold research question. These results propose that chronotherapeutic strategies such as SD, regardless of their long-term efficacy, may exist useful therapeutic tools to treat the symptoms of bipolar depression while the initial effect of antidepressant drugs is achieved. This finding is consistent with the widely documented rapid-onset antidepressant effects of SD (10, fourteen, 23). Information technology should exist considered that, even though drugs' antidepressant effects increment over a catamenia of weeks, the impact of chronotherapeutic strategies was non statistically significant at week 2 or further on. SD's utilise in the medium or long term would not be so well justified if pharmacological therapy is already effective. Furthermore, if mood medication shows a synergistic effect on brusque-term follow-up, at that place is no reason to utilise TSD without drugs. A possible exception would be patients who have an accented contraindication for mood medication utilise. Relative to the long-term effects of adding medication to TSD, such strategy suggests a much lower charge per unit of symptomatic relapse after recovery from sleep when TSD is used alone. Our results showed that concomitant use of mood medications and TSD maintained clinical remission on the long-term follow-up phase.
In our search, only ii previous meta-analysis related to SD were plant. The showtime (18) studied diurnal variation as a response predictor for SD. Recently some other meta-analysis was published including a full of 66 studies with a huge heterogeneity (different SD modalities, different depressive samples, demographics and different definitions of treatment response) and showed an overall response rate of 45% among the half dozen randomized studies and fifty% amidst the remaining nonrandomized included studies (24). Our results differed from this meta-analysis since they did not find a pregnant effect of calculation medications to SD. The fact that they included unipolar, bipolar, and mixed patient samples, and did not brand a departure between curt- and long-term outcomes might explain our different results. Moreover, the same meta-analysis ended that response charge per unit was non significantly affected by SD modality or sample blazon (unipolar, bipolar, or mixed). Unlike what has been reported in previous publications, the results of that meta-analysis indicate a nonsignificant tendency of a lower response in bipolar patients when compared to unipolar patients. This might exist explained by the exclusion of studies in which other chronotherapeutic strategies were practical in conjunction with SD that bear witness better response rates. For this reason, the authors propose that bipolar patients could benefit from SD mainly when it is associated with other chronotherapeutic therapies.
Contempo clinical guidelines in Major Depression Disorder (41), consider SD as a 3rd-line adjunctive therapy for more severe and refractory forms of MDD in combination with other chronotherapeutic strategies. On the other paw, bipolar disorder clinical guidelines (42), consider chronotherapeutic strategies every bit novel or experimental options and are therefore seen as the last step among therapeutic alternatives. A contempo systematic review claim that sleep deprivation research provides a low level of evidentiary support, mainly from uncontrolled studies, for the acute management of bipolar depression (43). Additionally, some promoters of TSD therapies advise its use even equally a first-line strategy for people affected by mood disorders, given its rubber (14). On the basis of our results, patients treated with TSD, with the concomitant use of mood medications (Lithium and monoaminergic drugs were used in the included trials) resulted in a rapid and sustained long-term antidepressant effect. TSD seems to be a recommended strategy in bipolar patients who fall into a depressive episode while keeping the previous treatment with mood medications such as lithium.
This report has some limitations and entails farther research questions and challenges. The get-go and probably major limitation is related to the small number of clinical trials available in literature that could meet inclusion criteria. It is important to annotation that, although there are many studies with available data most response rates and fifty-fifty relapse rates in patients subjected to unlike modalities of sleep deprivation, many of these accept dissimilar methodologies and objectives than those analyzed in our study, such as functional brain studies or analysis of biological markers in responder patients versus not-responders to sleep deprivation. However, for the purposes of our initial enquiry questions, clinical trials that straight assessed the impact of pharmacological and chronotherapeutic interventions in comparable terms were necessary. This inquiry feature limited the number of studies that encounter the inclusion criteria and showed that this is a research area in which new clinical trials are strongly needed to obtain more than conclusive results. This would allow for more informed clinical conclusion making. Some other limitation of this written report is related to the small number of patients that each selected clinical trial recruited. The very difficulties that come when implementing these treatment modalities could explicate low participant numbers. Recruiting volunteers or patients willing to receive these treatments tin can be challenging. Moreover, the included trials had differences in dependent and independent variables, such equally: differences in prescribed mood medications (different antidepressants and Lithium) and doses, consequence scales (VAS, HDRS, MADRS), and chronotherapeutic modalities (PSD, TSD with or without light therapy or other biological treatments), placebo conditions and diagnosis criteria. These differences must be considered as limitations for the generalization of our findings.
Although a new timeframe criterion for hypomania was added in DSM IV compared to DSM-III-R (44) (the 2 versions used in the trials included in this meta-analysis), this is probably a comparatively minor limiting factor. However, the evolution of the definition of bipolar disorder type I, type 2, and bipolar spectrum disorders is an issue to be considered in the heterogeneity of the samples in hereafter research.
These limitations could not simply significantly restrict the generalization of our findings, but they are also a phone call for researchers to consider lowering levels of heterogeneity in key methodological parameters such as randomization and placebo medication, despite the inherent difficulties of chronotherapy enquiry. Furthermore, trials accept not yet systematically addressed other important questions such equally the differential effect of TSD regarding bipolar subtypes.
Conclusion
Our results propose that adding chronotherapeutic interventions to mood medications for depressive episodes in bipolar patients seems to increase the antidepressant effect of the drugs early on of the treatment and maintaining the mood effects for the long-term follow-up.
Writer Contributions
JR-Thou, ER-S, PV, and LR conceived the written report. JR-Thousand and ER-Southward did the literature review, statistical analyses, and drafted the report. JR-K and ER-Southward extracted the data. All authors interpreted the data and edited and approved the final study.
Funding
Work by JR-Yard was funded by CONICYT PIA ACT 1414 and CONICYT FONDECYT (Ref: 3190311). PV has received salary support through the Fund for Innovation and Competitiveness (FIC) of the Chilean Ministry of Economy, Development and Tourism, through the Millennium Scientific Initiative (grant number IS130005). The cost of publication was funded by Fundacion Infirmary Clínico de la Universidad de Republic of chile.
Disharmonize of Involvement
The authors declare that the enquiry was conducted in the absence of whatsoever commercial or financial relationships that could be construed equally a potential conflict of interest.
Acknowledgments
The work was carried out at the Psychiatric Clinic of the University of Chile. We admit psychiatrists, psychologists, and residents of the Psychiatric Clinic of the University of Chile for their comments and suggestions. We thank Luz Maria Alliende for his insightful comments on the manuscript.
Supplementary Material
The Supplementary Material for this article tin can be found online at: https://www.frontiersin.org/articles/10.3389/fpsyt.2020.00070/full#supplementary-material
References
1. Charlson F, van OM, Flaxman A, Cornett J, Whiteford H, Saxena S. New WHO prevalence estimates of mental disorders in conflict settings: a systematic review and meta-analysis. Lancet (2019) 394(10194):240–8. doi: 10.1016/S0140-6736(19)30934-i
PubMed Abstract | CrossRef Total Text | Google Scholar
2. Baldessarini RJ, Salvatore P, Khalsa H-MK, Gebre-Medhin P, Imaz H, González-Pinto A, et al. Morbidity in 303 first-episode bipolar I disorder patients. Bipolar Disord (2010) 12(iii):264–lxx. doi: 10.1111/j.1399-5618.2010.00812.x
PubMed Abstract | CrossRef Full Text | Google Scholar
iii. Bonnín CDM, Reinares M, Martínez-Arán A, Jiménez East, Sánchez-Moreno J, Solé B, et al. Improving operation, quality of life, and well-being in patients with bipolar disorder. Int J Neuropsychopharmacol (2019) 22(8):467–77. doi: 10.1093/ijnp/pyz018
PubMed Abstract | CrossRef Full Text | Google Scholar
5. McGirr A, Vöhringer PA, Ghaemi SN, Lam RW, Yatham LN. Prophylactic and efficacy of adjunctive second-generation antidepressant therapy with a mood stabiliser or an atypical antipsychotic in astute bipolar depression: a systematic review and meta-assay of randomised placebo-controlled trials. Lancet Psychiatry (2016) iii(12):1138–46. doi: 10.1016/S2215-0366(xvi)30264-4
PubMed Abstract | CrossRef Full Text | Google Scholar
7. Crump C, Sundquist K, Winkleby MA, Sundquist J. Comorbidities and mortality in bipolar disorder: a Swedish National Cohort Study. JAMA Psychiatry (2013) 70(9):931–ix. doi: x.1001/jamapsychiatry.2013.1394
PubMed Abstract | CrossRef Full Text | Google Scholar
eight. Schulte W. Zum Priblem der provokation und kupierung von Melancholischen Phasen. Schweizer. Arch Neurol Neurochem Psychiatr (1971) 109:427–35.
Google Scholar
ix. Steinberg H, Hegerl U. Johann Christian Baronial Heinroth on slumber deprivation as a therapeutic option for depressive disorders. Sleep Med (2014) 15(9):1159–64. doi: x.1016/j.sleep.2014.03.027
PubMed Abstract | CrossRef Full Text | Google Scholar
11. Wu JC, Kelsoe JR, Schachat C, Bunney BG, DeModena A, Golshan Due south, et al. Rapid and sustained antidepressant response with slumber deprivation and chronotherapy in bipolar disorder. Biol Psychiatry (2009) 66(3):298–301. doi: 10.1016/j.biopsych.2009.02.018
PubMed Abstract | CrossRef Full Text | Google Scholar
12. Benedetti F. Antidepressant chronotherapeutics for bipolar low. Dialog Clin Neurosci (2012) 14(four):401–11.
Google Scholar
xv. Riemann D, König A, Hohagen F, Kiemen A, Voderholzer U, Backhaus J, et al. How to preserve the antidepressive result of sleep deprivation: A comparison of slumber phase accelerate and slumber phase delay. Eur Arch Psychiatry Clin Neurosci (1999) 249:231–7. doi: 10.1007/s004060050092
PubMed Abstruse | CrossRef Full Text | Google Scholar
17. Barbini B, Colombo C, Benedetti F, Campori E, Bellodi L, Smeraldi Due east. The unipolar–bipolar dichotomy and the response to sleep deprivation. Psychiatry Res (1998) 79(one):43–l. doi: ten.1016/S0165-1781(98)00020-1
PubMed Abstract | CrossRef Full Text | Google Scholar
19. Benedetti F, Lucca A, Brambilla F, Colombo C, Smeraldi E. Interleukine-vi serum levels correlate with response to antidepressant sleep deprivation and slumber phase accelerate. Prog Neuropsychopharmacol Biol Psychiatry (2002) 26(6):1167–lxx. doi: 10.1016/S0278-5846(02)00255-v
PubMed Abstract | CrossRef Full Text | Google Scholar
23. Wu JC, Bunney Nosotros. The biological footing of an antidepressant response to slumber deprivation and relapse: review and hypothesis. Am J Psychiatry (1990) 147(1):fourteen–21. doi: ten.1176/ajp.147.1.14
PubMed Abstract | CrossRef Total Text | Google Scholar
24. Boland EM, Rao H, Dinges DF, Smith RV, Goel N, Detre JA, et al. Meta-analysis of the antidepressant effects of acute sleep deprivation. J Clin Psychiatry (2017) 78(8):e1020–34. doi: 10.4088/JCP.16r11332
PubMed Abstract | CrossRef Full Text | Google Scholar
25. Benedetti F, Barbini B, Lucca A, Campori East, Colombo C, Smeraldi East. Sleep deprivation hastens the antidepressant activity of fluoxetine. Eur Arch Psychiatry Clin Neurosci (1997) 247(2):100–3. doi: 10.1007/BF02900200
PubMed Abstract | CrossRef Total Text | Google Scholar
26. Benedetti F, Colombo C, Barbini B, Campori E, Smeraldi E. Ongoing lithium treatment prevents relapse afterward total sleep deprivation. J Clin Psychopharmacol (1999) nineteen(three):240–5. doi: 10.1097/00004714-199906000-00007
PubMed Abstract | CrossRef Full Text | Google Scholar
27. Smeraldi E, Benedetti F, Barbini B, Campori East, Colombo C. Sustained antidepressant effect of sleep deprivation combined with pindolol in bipolar depression. a placebo-controlled trial. Neuropsychopharmacology> (1999) 20(4):380–5. doi: 10.1016/S0893-133X(98)00129-8
PubMed Abstract | CrossRef Full Text | Google Scholar
28. Benedetti F, Barbini B, Campori Due east, Fulgosi MC, Pontiggia A, Colombo C. Sleep phase advance and lithium to sustain the antidepressant effect of full slumber deprivation in bipolar low: new findings supporting the internal coincidence model? J Psychiatr Res (2001) 35(6):323–ix. doi: ten.1016/S0022-3956(01)00034-half-dozen
PubMed Abstract | CrossRef Full Text | Google Scholar
29. Benedetti F, Campori E, Barbini B, Fulgosi MC, Colombo C. Dopaminergic augmentation of sleep impecuniousness effects in bipolar low. Psychiatry Res (2001) 104(3):239–46. doi: 10.1016/S0165-1781(01)00332-viii
PubMed Abstract | CrossRef Total Text | Google Scholar
thirty. Colombo C, Lucca A, Benedetti F, Barbini B, Campori E, Smeraldi Due east. Total sleep deprivation combined with lithium and light therapy in the treatment of bipolar depression: replication of main effects and interaction. Psychiatry Res (2000) 95(i):43–53. doi: x.1016/S0165-1781(00)00164-v
PubMed Abstract | CrossRef Total Text | Google Scholar
35. Fergusson D, Aaron SD, Guyatt One thousand, Hébert P. Post-randomisation exclusions: the intention to treat principle and excluding patients from analysis. BMJ (2002) 325(7365):652–4. doi: 10.1136/bmj.325.7365.652
PubMed Abstract | CrossRef Full Text | Google Scholar
36. Borenstein. Introduction to Meta-Analysis. Edición: i. Chichester: John Wiley & Sons (2009). 456 p.
Google Scholar
37. Higgins JPT, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman Advertizing, et al. The cochrane collaboration's tool for assessing risk of bias in randomised trials. BMJ (2011) 343. doi: 10.1136/bmj.d5928.
CrossRef Full Text | Google Scholar
38. Baxter LR, Liston EH, Schwartz JM, Altshuler LL, Wilkins JN, Richeimer S, et al. Prolongation of the antidepressant response to partial sleep impecuniousness by lithium. Psychiatry Res (1986) 19(1):17–23. doi: 10.1016/0165-1781(86)90088-0
PubMed Abstract | CrossRef Full Text | Google Scholar
39. Leibenluft E, Moul DE, Schwartz PJ, Madden PA, Wehr TA. A clinical trial of sleep deprivation in combination with antidepressant medication. Psychiatry Res (1993) 46(3):213–27. doi: 10.1016/0165-1781(93)90090-4
PubMed Abstract | CrossRef Full Text | Google Scholar
40. Suzuki M, Dallaspezia S, Locatelli C, Uchiyama M, Colombo C, Benedetti F. Does early response predict subsequent remission in bipolar low treated with repeated sleep deprivation combined with light therapy and lithium? J Affect Disord (2018) 229:371–6. doi: ten.1016/j.jad.2017.12.066
PubMed Abstract | CrossRef Total Text | Google Scholar
41. Ravindran AV, Balneaves LG, Faulkner M, Ortiz A, McIntosh D, Morehouse RL, et al. Canadian Network for Mood and Feet Treatments (CANMAT) 2016 clinical guidelines for the direction of adults with major depressive disorder: department 5. complementary and alternative medicine treatments. Tin can J Psychiatry (2016) 61(nine):576–87.
PubMed Abstract | Google Scholar
42. Yatham LN, Kennedy SH, Parikh SV, Schaffer A, Beaulieu S, Alda M, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) collaborative update of CANMAT guidelines for the direction of patients with bipolar disorder: update 2013. Bipolar Disord (2013) xv(1):1–44. doi: 10.1111/bdi.12025
PubMed Abstruse | CrossRef Full Text | Google Scholar
43. Gottlieb JF, Benedetti F, Geoffroy PA, Henriksen TEG, Lam RW, Murray Thou, et al. The chronotherapeutic treatment of bipolar disorders: a systematic review and practice recommendations from the ISBD task force on chronotherapy and chronobiology. Bipolar Disord (2019) 21(8):741–73. doi: ten.1111/bdi.12847
PubMed Abstract | CrossRef Full Text | Google Scholar
44. Bricklayer BL, Chocolate-brown ES, Croarkin PE. Historical underpinnings of bipolar disorder diagnostic criteria. Behav Sci (2016) six(3):14. doi: 10.3390/bs6030014
CrossRef Total Text | Google Scholar
Source: https://www.frontiersin.org/articles/10.3389/fpsyt.2020.00070/full
0 Response to "Sleep Medication Reviews for People With Bi Polar"
Postar um comentário